The Pivotal Role of Pharmaceutical Intermediates in Modern Drug Synthesis
The pharmaceutical industry relies heavily on a complex ecosystem of specialized chemicals to develop life-saving and life-improving medications. Central to this process are pharma intermediates, critical compounds formed during the synthesis of Active Pharmaceutical Ingredients (APIs). These intermediates are not the final drug products themselves but are essential building blocks that undergo further chemical reactions and purification steps. Their quality, consistency, and efficient production directly impact the safety, efficacy, and cost-effectiveness of the final pharmaceutical product. This article delves into the technical aspects, market dynamics, and strategic considerations surrounding these vital chemical entities, with a particular focus on high-purity compounds like N,N’-Dimethyl Urea and its diverse applications.
The precision required in pharmaceutical manufacturing demands intermediates with exceptionally high purity and well-defined chemical characteristics. This includes rigorous control over stereochemistry, impurity profiles, and physical properties, all of which are paramount for regulatory compliance and therapeutic success.
Manufacturing Process of Key Pharma Intermediates
The production of pharma intermediates is a multi-step, highly controlled process designed to achieve specific chemical structures with minimal impurities. Taking N,N’-Dimethyl Urea as an illustrative example, its synthesis typically involves the reaction of urea with methanol or through the methylation of urea. The process flow for such an intermediate often follows a sequence of synthesis, purification, and quality assurance.
Process Flow Diagram: N,N’-Dimethyl Urea Synthesis
Step 1: Raw Material Preparation
- ➤ High-purity Urea (C(NH2)2O)
- ➤ Methanol (CH3OH) / Methylamine (CH3NH2)
- ➤ Catalyst (if required)
Step 2: Reaction & Synthesis
- ➤ Reactants are charged into a jacketed reactor under controlled temperature and pressure.
- ➤ The methylation reaction proceeds, forming N,N’-Dimethyl Urea (DMU).
- ➤ By-product removal (e.g., ammonia or water).
Step 3: Purification & Isolation
- ➤ Filtration: Removal of insoluble impurities.
- ➤ Crystallization: Dissolving the crude product and recrystallizing to enhance purity.
- ➤ Drying: Removal of residual solvents to achieve desired moisture content.
Step 4: Quality Control & Packaging
- ➤ Comprehensive testing against established specifications (e.g., purity, melting point, assay).
- ➤ Packaging in compliant container111s under inert atmosphere to maintain stability.
Testing Standards & Quality Assurance: All stages are governed by stringent quality management systems such as ISO 9001 and Good Manufacturing Practices (GMP) guidelines. Analytical methods like HPLC, GC, NMR, and Mass Spectrometry are routinely employed to ensure the identity, purity, and absence of critical impurities.
Target Industries: These pharma intermediates primarily serve the pharmaceutical and fine chemical industries. For instance, N,N’-Dimethyl Urea finds applications beyond pharmaceuticals, including as a precursor in organic synthesis and as an intermediate for certain agrochemicals.
Advantages: The precise control over the manufacturing process ensures a product with high purity (>99.5%), excellent batch-to-batch consistency, and extended service life due to its stability under specified storage conditions. This translates into energy savings for end-users by reducing the need for further purification steps and providing reliable yields in subsequent reactions, while minimizing waste and ensuring corrosion resistance of processing equipment.

Figure 1: Advanced manufacturing facility for specialized chemical intermediates.
Industry Trends and Market Dynamics for Pharma Intermediates
The market for pharma intermediates is shaped by several dynamic trends. A significant driver is the increasing demand for novel APIs due to a growing global population and rising prevalence of chronic diseases. This fuels innovation in drug discovery, which in turn necessitates a wider array of sophisticated intermediates.
- Sustainable Chemistry: There’s a growing emphasis on green chemistry principles, pushing for eco-friendly synthesis routes, reduced solvent usage, and minimization of hazardous waste. Manufacturers are investing in biocatalysis and flow chemistry to meet these demands.
- Supply Chain Resilience: Recent global events have highlighted the critical need for robust and diversified supply chains. Pharmaceutical companies are seeking reliable suppliers with strong quality control and geopolitical stability, sometimes leading to regionalization of production.
- Advanced Analytical Techniques: Continuous improvements in analytical instrumentation allow for more precise characterization and impurity profiling of intermediates, ensuring compliance with increasingly stringent regulatory requirements from agencies like the FDA and EMA.
- Digitalization & AI: The adoption of AI and machine learning in chemical synthesis is optimizing reaction conditions, predicting yields, and accelerating the discovery of new synthetic pathways for intermediates.
- Custom Synthesis & CDMOs: The trend towards outsourcing R&D and manufacturing to Contract Development and Manufacturing Organizations (CDMOs) continues, requiring specialized expertise in custom synthesis of complex intermediates.
These trends underscore the need for manufacturers of pharma intermediates to be agile, innovative, and committed to the highest standards of quality and sustainability.
Technical Specifications: Refine N,N’-Dimethyl Urea
Our Refine N,N’-Dimethyl Urea is manufactured to exacting standards, ensuring high purity and consistent performance for demanding applications. Below are the key technical specifications for this critical intermediate.
Product Specification Table: Refine N,N’-Dimethyl Urea
| Parameter | Specification | Method |
|---|---|---|
| Chemical Name | N,N’-Dimethylurea | — |
| CAS No. | 13032-26-3 | — |
| Molecular Formula | C3H8N2O | — |
| Molecular Weight | 88.11 g/mol | — |
| Purity (HPLC) | ≥ 99.5% | HPLC |
| Appearance | White Crystalline Powder | Visual |
| Melting Point | 105 – 107 °C | USP <741> |
| Loss on Drying | ≤ 0.5% | USP <731> |
| Heavy Metals (as Pb) | ≤ 10 ppm | USP <231> |
| Related Substances | ≤ 0.3% (Total) | HPLC |
These specifications are crucial for pharmaceutical manufacturers to ensure their raw materials meet the stringent requirements for API synthesis, contributing to the overall quality and safety of the final drug product.
Application Scenarios and Technical Advantages
The versatility of high-quality chemical intermediates like N,N’-Dimethyl Urea extends across various critical applications, particularly within the pharmaceutical and polymer industries.
Key Application Areas:
- Pharmaceutical Synthesis: N,N’-Dimethyl Urea is a vital building block in the synthesis of various pharmaceutical compounds. It serves as a carbamoylation agent or a nitrogen source in reactions, enabling the creation of complex heterocyclic structures common in modern APIs. Its high purity ensures minimal contamination in subsequent drug synthesis steps, which is critical for achieving target API specifications and reducing downstream purification costs.
- Chemical Synthesis: Beyond pharmaceuticals, it is used as an intermediate in the production of other fine chemicals, agrochemicals, and specialty polymers. Its well-defined reactivity makes it a preferred reagent for formylation and other synthetic transformations.
- Additives for Polymers: Dimethylurea can also function as a formaldehyde scavenger, particularly in urea-formaldehyde resins. This property is crucial in reducing formaldehyde emissions from composite wood products, aligning with stricter environmental regulations. As an additive for polymers, it contributes to improved material properties or acts as a processing aid, demonstrating its broad industrial utility.
Technical Advantages:
- Superior Purity: Our pharma intermediates boast purity levels exceeding 99.5%, minimizing side reactions and ensuring high yields in downstream processes. This significantly reduces the need for extensive purification of final products, translating to operational efficiencies and cost savings.
- Consistent Batch Quality: Rigorous quality control protocols, including comprehensive analytical testing for every batch, guarantee exceptional consistency. This reliability is paramount for pharmaceutical manufacturing, where batch-to-batch variation can lead to significant production delays and regulatory challenges.
- Optimized Reactivity: The controlled chemical profile of our intermediates ensures predictable and efficient reactivity, allowing for streamlined synthesis pathways and improved process control for our clients.
- Enhanced Stability: Our products are designed for stability, with optimal packaging and storage conditions specified to maintain chemical integrity over their intended service life, reducing degradation and ensuring long-term usability.
- Regulatory Compliance: Manufactured under strict adherence to international standards (e.g., ISO, GMP principles), our intermediates help clients meet stringent regulatory requirements for their end products, facilitating faster market approval.
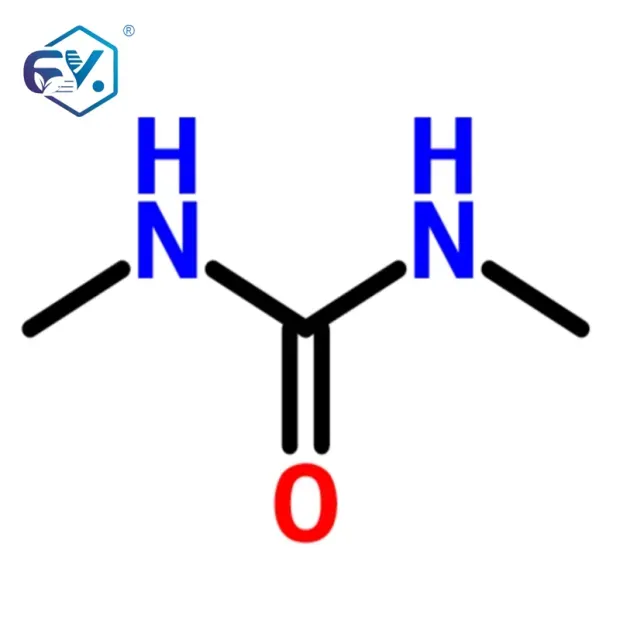
Figure 2: Quality control laboratory ensuring purity and consistency of chemical intermediates.
Vendor Comparison and Customization Solutions
Selecting the right vendor for pharma intermediates is a critical strategic decision that impacts product quality, supply chain reliability, and overall project timelines. While many suppliers exist, a discerning approach is necessary to ensure long-term success.
Key Factors for Vendor Evaluation:
| Factor | High-Quality Vendor Profile | Risk for Substandard Vendors |
|---|---|---|
| Quality & Purity | GMP compliance, >99.5% purity, robust analytical data (CoA). | Inconsistent purity, undisclosed impurities, lack of comprehensive CoA. |
| Regulatory Compliance | ISO 9001, strong regulatory support, audit-ready facilities. | Weak documentation, non-existent QMS, potential for regulatory rejections. |
| Supply Chain Stability | Multiple production sites, strong logistics, transparent lead times. | Single-source dependency, unpredictable delays, opaque communication. |
| Technical Support | Experienced R&D team, problem-solving expertise, custom synthesis capabilities. | Limited technical expertise, inability to resolve issues, no customization. |
| Pricing Structure | Transparent, competitive, value-driven pricing, long-term contracts. | Hidden costs, frequent price fluctuations, high minimum order quantities. |
Customized Solutions:
Recognizing that every pharmaceutical synthesis project has unique requirements, we offer extensive customization capabilities for our pharma intermediates.
- Tailored Purity Profiles: While standard products meet high purity, we can fine-tune purification processes to meet specific impurity limits or enantiomeric excess requirements for highly sensitive applications.
- Custom Packaging & Sizes: From bulk quantities to specialized packaging for aseptic environments, we adapt to client logistical and operational needs.
- Synthesis Route Development: Our R&D team can collaborate on developing novel or optimized synthesis routes for intermediates, enhancing efficiency, reducing costs, or addressing specific intellectual property concerns.
- Formulation Modifications: For applications such as additives for polymers, we can assist in modifying the physical form or solvent compatibility to better integrate with specific polymer systems.
Our commitment to flexibility and technical partnership ensures that clients receive intermediates perfectly aligned with their project specifications and strategic goals.
Application Case Studies
These case studies illustrate how our high-quality pharma intermediates provide tangible benefits and solutions for our partners.
Case Study 1: Streamlining API Synthesis for a Cardiovascular Drug
A leading pharmaceutical client faced challenges in synthesizing a key cardiovascular API. The existing supply of a critical intermediate had inconsistent purity, leading to variable yields and increased purification steps in their final API production. Our team provided Refine N,N’-Dimethyl Urea with a guaranteed purity of ≥ 99.8% and a detailed impurity profile. By integrating our high-purity intermediate, the client observed a 15% increase in API yield, a 20% reduction in purification cycle time, and a significant decrease in off-spec batches. This direct impact on process efficiency and product quality showcased the value of a superior quality intermediate in complex API manufacturing.
Case Study 2: Enhancing Polymer Performance with Specialized Additives
An industrial client specializing in polymer manufacturing for building materials sought to reduce formaldehyde emissions from their urea-formaldehyde resins to meet new European environmental standards. Traditional formaldehyde scavengers proved either too costly or ineffective at low concentrations. We collaborated with their R&D team to develop a modified dimethylurea derivative, tailored for their specific resin system. This customized solution, provided as an additive for polymers, achieved a 40% reduction in formaldehyde emissions at a significantly lower cost compared to existing solutions, leading to full compliance and a competitive advantage in the market.
Frequently Asked Questions (FAQ)
Q1: What certifications do your pharma intermediates products hold?
A1: Our manufacturing facilities operate under strict ISO 9001 certified Quality Management Systems. Products intended for pharmaceutical use adhere to GMP (Good Manufacturing Practices) principles, and we provide comprehensive Certificates of Analysis (CoA) for every batch, confirming compliance with specified parameters.
Q2: What is the typical lead time for an order of N,N’-Dimethyl Urea?
A2: Standard lead times for our Refine N,N’-Dimethyl Urea generally range from 2-4 weeks, depending on order quantity and current inventory levels. For custom synthesis or large-volume orders, lead times are discussed and agreed upon during the quotation phase, with transparency at every step. We maintain strategic inventory levels to support just-in-time delivery for our key partners.
Q3: Do you offer samples for R&D purposes?
A3: Yes, we understand the importance of initial testing. We offer evaluation samples for R&D and pilot-scale projects. Please contact our sales team with your specific requirements, and we will arrange for sample delivery.
Q4: What are your warranty and after-sales support commitments?
A4: We provide a comprehensive warranty on all our products, guaranteeing them to meet the agreed-upon specifications and quality standards upon delivery. Our dedicated after-sales support team is available to assist with any technical inquiries, troubleshooting, or quality concerns that may arise, ensuring continued client satisfaction and product performance.
Contact and Collaboration
For further technical specifications, custom synthesis inquiries, or to discuss your specific pharma intermediates needs, please reach out to our expert team. We are committed to fostering long-term partnerships built on trust, quality, and mutual success.
Contact us at: info@hbgxchemical.com or visit our website for more details.
Hebei Guangxing Chemical Co., Ltd. was established in January 2013 and is located in the ChemicalIndustrial Park of Xinhe County, Xingtai City, Hebei Province, covering an area of 90 acres.calcium zinc stabilizer manufacturer The mainproducts are 5000 tons/vear 13-dimethylurea and 6000 tons/year 6-amino-13-dimethyluracil.Hebei Guangxing Chemical Co., Ltd. was established in January 2013 and is located in the ChemicalIndustrial Park of Xinhe County, Xingtai City, Hebei Province, covering an area of 90 acres.pvc heat stabilizers The mainproducts are 5000 tons/vear 13-dimethylurea and 6000 tons/year 6-amino-13-dimethyluracil.uracils|super blog