Introduction to Atovaquone: A Critical API in Pharmaceutical Development
In the highly specialized field of pharmaceutical active pharmaceutical ingredients (APIs), the demand for compounds with proven efficacy and robust manufacturing integrity is paramount. Atovaquone, a hydroxynaphthoquinone derivative, stands as a pivotal antiparasitic and antiprotozoal agent. Its broad-spectrum activity against organisms such as Pneumocystis jirovecii (formerly P. carinii) and various malaria parasites, including Plasmodium falciparum, positions it as an essential component in global health initiatives. This detailed exposition delves into the technical aspects, application nuances, and market dynamics surrounding this critical API, tailored for B2B decision-makers and technical procurement specialists.
Our commitment to quality and technical excellence ensures the delivery of Atovaquone meeting the most stringent pharmacopoeial standards, facilitating its integration into life-saving drug formulations.
Manufacturing Process Flow of Atovaquone
The synthesis of Atovaquone is a complex, multi-step organic chemistry process, demanding precise control over reaction conditions and purification techniques to ensure high yield and exceptional purity. Our manufacturing facility adheres to cGMP (current Good Manufacturing Practices) guidelines, employing state-of-the-art equipment and validated protocols.
Key Process Steps (Schematic):
- Raw Material Sourcing & Pre-treatment: High-purity 2-hydroxy-1,4-naphthoquinone derivatives and alkylating agents are meticulously selected. Materials undergo initial quality checks for identity, purity, and absence of contaminants, ensuring adherence to pharmacopoeial specifications before entering production.
- Reaction & Synthesis: The core synthesis involves multi-stage chemical reactions, typically including a key alkylation step and subsequent cyclization/oxidation. These reactions are carried out in controlled reactor environments, often under inert atmosphere to prevent undesired side reactions. Critical process parameters (CPPs) such as temperature, pressure, stirring speed, and reactant addition rates are continuously monitored.
- Purification & Isolation: Following synthesis, the crude product undergoes rigorous purification. This often involves crystallization, solvent extraction, and precipitation methods. Multiple rounds of purification may be necessary to remove impurities, unreacted starting materials, and byproducts, achieving the desired purity profile.
- Drying & Milling: The purified wet cake is then dried using vacuum dryers or fluid bed dryers to achieve the specified moisture content. Depending on downstream application requirements, the dried API may be milled or micronized to obtain a specific particle size distribution (PSD), which is crucial for dissolution and bioavailability.
- Quality Control & Packaging: Every batch undergoes extensive analytical testing to confirm identity, purity, assay, residual solvents, heavy metals, and other critical quality attributes (CQAs) against pharmacopoeial standards (e.g., USP, EP, BP). Once approved, the API is carefully packaged in sealed, pharmaceutical-grade container111s under controlled environmental conditions to maintain stability and prevent contamination during storage and transport.
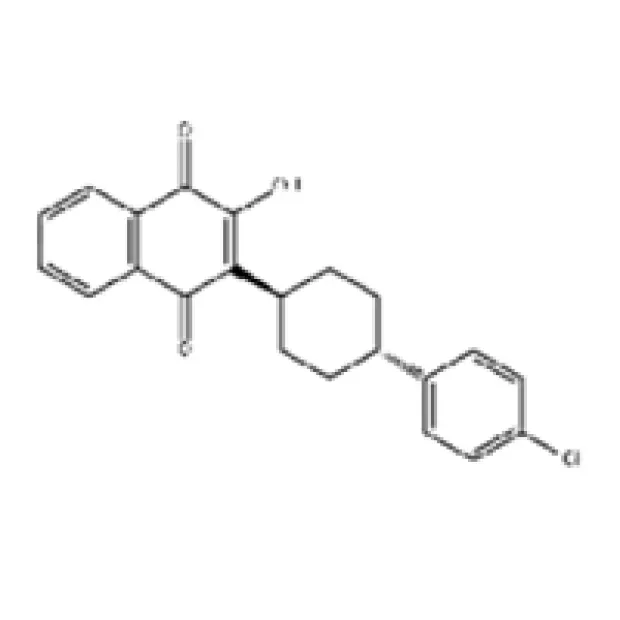
Figure 1: Conceptual diagram of a typical pharmaceutical API synthesis and purification workflow.
Testing Standards: Our Atovaquone production adheres strictly to international pharmaceutical standards, including USP (United States Pharmacopeia), EP (European Pharmacopoeia), and BP (British Pharmacopoeia). Compliance with ISO 9001 for quality management systems and ISO 14001 for environmental management systems further underscores our commitment to excellence.
Service Life & Stability: The shelf life of Atovaquone API, when stored under recommended conditions (typically cool, dry, and protected from light), is typically 2-3 years, supported by stability studies conducted according to ICH guidelines. This extended stability ensures flexibility in formulation and manufacturing schedules for our clients.
Target Industries: The primary target industries for Atovaquone are pharmaceutical manufacturers, contract research organizations (CROs), and generic drug developers focusing on antiparasitic and antiprotozoal medications. Its applications span various therapeutic areas within infectious diseases.
Industry Trends and Market Dynamics for Atovaquone
The market for anti-infective APIs like Atovaquone is influenced by several key trends. The increasing prevalence of drug-resistant pathogens, particularly in malaria, drives the need for effective alternative treatments. The emergence of co-infection scenarios, such as HIV and opportunistic infections like Pneumocystis jirovecii pneumonia (PCP), sustains demand for therapeutics like Atovaquone, which is a cornerstone in PCP prophylaxis and treatment.
- Global Health Initiatives: Programs by organizations such as the WHO and various national health bodies continue to support the distribution and accessibility of essential medicines, including those containing Atovaquone, in endemic regions.
- Generics Market Expansion: As patents expire for innovator drugs, the generic API market experiences robust growth, driving competition and innovation in cost-effective manufacturing without compromising quality.
- Supply Chain Resilience: Post-pandemic, there’s an increased focus on diversifying API sourcing and strengthening supply chain resilience to ensure uninterrupted availability of critical drugs.
- Regulatory Harmonization: Efforts towards global regulatory harmonization (e.g., ICH guidelines) facilitate smoother market entry and ensure consistent quality standards across different regions.
These trends underscore the importance of reliable, high-quality API suppliers capable of meeting stringent regulatory and market demands for Atovaquone.
Technical Specifications and Parameters of Atovaquone
Understanding the precise technical specifications of Atovaquone is crucial for formulation development and quality assurance. Our product adheres to the following critical parameters, verified through comprehensive analytical testing.
Product Specification Table: Atovaquone API
| Parameter | Specification (USP/EP Grade) | Methodology |
|---|---|---|
| CAS Number | 95233-18-4 | N/A |
| Chemical Formula | C22H19ClO3 | N/A |
| Molecular Weight | 366.84 g/mol | N/A |
| Appearance | Yellow to Orange Crystalline Powder | Visual Inspection |
| Assay (HPLC, Anhydrous) | 98.0% – 102.0% | USP/EP Monograph |
| Related Substances (Individual) | NMT 0.10% | HPLC |
| Total Related Substances | NMT 0.50% | HPLC |
| Water Content | NMT 0.5% | Karl Fischer Titration |
| Residual Solvents | Meets ICH Class 2 & 3 limits | GC-HS |
| Heavy Metals | NMT 10 ppm | ICP-OES/AAS |
| Particle Size Distribution (PSD) | Customizable per client specification | Laser Diffraction |
NMT = Not More Than; HPLC = High-Performance Liquid Chromatography; GC-HS = Gas Chromatography-Headspace; ICP-OES = Inductively Coupled Plasma – Optical Emission Spectrometry; AAS = Atomic Absorption Spectroscopy.
Application Scenarios of Atovaquone
Atovaquone exhibits versatile efficacy across various challenging infectious disease indications. Its mechanism of action, involving inhibition of the parasite’s mitochondrial electron transport chain, makes it particularly effective where other agents may face resistance.
- Malaria Treatment and Prophylaxis: Primarily used in combination with Proguanil (Malarone®) for the prophylaxis and treatment of malaria caused by Plasmodium falciparum, including chloroquine-resistant strains. It is particularly valuable for travelers to endemic areas and in regions with high drug resistance.
- Pneumocystis Pneumonia (PCP) Treatment: Atovaquone is an established alternative for the treatment of mild to moderate PCP in individuals who are intolerant to trimethoprim-sulfamethoxazole (TMP-SMX), especially in HIV-infected patients.
- PCP Prophylaxis: It is also indicated for prophylaxis against PCP in high-risk individuals, such as those with advanced HIV disease, where TMP-SMX is contraindicated or not tolerated.
- Toxoplasmosis: While not a first-line agent, Atovaquone has shown activity against Toxoplasma gondii and is sometimes considered in specific clinical scenarios, particularly in immunocompromised patients.
- Babesiosis: Used off-label or in combination with azithromycin for the treatment of babesiosis, a tick-borne parasitic disease.
The robust clinical profile of Atovaquone ensures its continued importance in managing these challenging infectious diseases globally.
Technical Advantages of Our Atovaquone
Our Atovaquone API offers distinct technical advantages derived from our optimized synthesis routes, stringent quality control, and advanced manufacturing capabilities.
- High Purity Profile: Achieved through multi-stage purification, our Atovaquone consistently meets or exceeds pharmacopoeial requirements for purity, with minimal related substances, critical for patient safety and drug efficacy.
- Optimized Particle Size Distribution (PSD): We offer customized PSD to enhance bioavailability and dissolution rates, crucial for oral formulations. This optimization reduces variability in drug absorption, leading to more predictable therapeutic outcomes.
- Batch-to-Batch Consistency: Our robust Quality Management System (QMS) and process validation ensure exceptional batch-to-batch consistency, reducing formulation challenges and accelerating regulatory approval for our clients.
- Regulatory Documentation Support: We provide comprehensive regulatory documentation, including Drug Master Files (DMFs) and Certificate of Suitability (CEP/COS), facilitating global market access for your finished products.
- Strong Supply Chain: With diversified raw material sourcing and strategic logistics, we offer a resilient and reliable supply of Atovaquone, mitigating supply risks for our pharmaceutical partners.
These advantages translate into tangible benefits for our B2B partners, enabling them to develop high-quality, effective, and compliant pharmaceutical products.
Vendor Comparison: Why Choose Our Atovaquone
Selecting the right API vendor is a critical strategic decision. This comparison highlights our differentiation in the highly competitive Atovaquone market.
Atovaquone Vendor Comparison
| Feature | Our Offering | Typical Competitor |
|---|---|---|
| Purity (Assay) | >99.5% (USP/EP Grade) | 98.0% – 99.0% |
| Related Substances | Total <0.2% | Total <0.5% |
| Regulatory Support | Complete DMF (Type II, US), CEP/COS, comprehensive technical dossier. Dedicated regulatory team. | Basic DMF or limited documentation. |
| Quality Certifications | cGMP, ISO 9001, ISO 14001. Regular audits by international agencies. | Basic GMP; fewer external audits. |
| Customization Options | Particle size, polymorphic form, specific excipient compatibility studies, custom packaging. | Standard product offerings only. |
| Lead Time | Consistent, reliable, typically 4-6 weeks for standard orders. | Variable, often longer lead times. |
Our dedication to superior quality, regulatory excellence, and customer-centric service positions us as a preferred partner for Atovaquone.
Customized Solutions for Atovaquone
Recognizing that formulation needs vary, we offer tailored solutions for Atovaquone to integrate seamlessly into diverse drug products. Our R&D and manufacturing teams are equipped to collaborate on specific requirements.
- Particle Size Engineering: We can customize the particle size distribution of Atovaquone through controlled milling and micronization techniques to optimize dissolution rates and bioavailability for specific oral dosage forms (e.g., tablets, suspensions).
- Polymorphic Control: Consistent polymorphic form is critical for API performance. We ensure control over the crystalline form of Atovaquone to guarantee stability and prevent unforeseen issues during formulation and storage.
- Custom Packaging: From bulk drums to smaller, specific-weight container111s, we offer packaging solutions tailored to client manufacturing processes and regulatory requirements, including specific material compatibility.
- Analytical Method Development & Validation: Our analytical laboratories can assist with developing and validating methods specific to your formulation needs or provide support for tech transfer processes.
- Impurity Profiling: We offer advanced impurity profiling and genotoxic impurity assessments to support new drug applications and meet stringent regulatory expectations.
Our flexibility and technical prowess enable us to be more than just a supplier; we are a strategic development partner.
Application Case Studies & Customer Feedback
Our Atovaquone has been successfully integrated into various pharmaceutical products by leading global manufacturers, demonstrating its reliability and quality in real-world applications.
Case Study 1: Enhanced Bioavailability in Malaria Prophylaxis
A major pharmaceutical client required Atovaquone with a highly consistent and optimized particle size for a novel co-formulation aimed at improving bioavailability and reducing dose frequency in malaria prophylaxis. Collaborating closely with their formulation team, we engineered Atovaquone API to a specific micronized grade, achieving a D90 < 5 µm with tight control over the particle size distribution. This resulted in a significantly improved dissolution profile and consistent absorption in their final drug product, leading to successful Phase III trials and subsequent market approval in several high-burden regions.
Case Study 2: Rapid Market Entry for Generic PCP Treatment
A generics manufacturer sought to rapidly launch an Atovaquone-based product for PCP treatment in a highly regulated market. Our comprehensive DMF (Drug Master File) and direct support from our regulatory affairs team significantly streamlined their regulatory submission process. Our API’s consistent high purity and stability data, backed by extensive stability studies, minimized their in-house testing burden and accelerated their time-to-market. The client successfully launched their product within 18 months, attributing a substantial portion of their success to the quality of our API and regulatory support.
Customer Feedback:
“The consistent quality of your Atovaquone has been instrumental in maintaining the integrity and efficacy of our formulations. Your technical support team is highly responsive and knowledgeable, making collaboration effortless. We consider your company a cornerstone of our API supply chain.” – VP of Manufacturing, Global Pharmaceutical Company.
Trustworthiness & Support
Frequently Asked Questions (FAQ)
- Q: What regulatory documentation is available for Atovaquone?
- A: We provide comprehensive documentation including a US DMF (Type II), CEP/COS, and a detailed technical package to support your regulatory submissions in major markets.
- Q: Can you provide samples for R&D purposes?
- A: Yes, we offer samples of Atovaquone for R&D and preliminary testing. Please contact our sales team to discuss your specific sample requirements.
- Q: What are the recommended storage conditions for Atovaquone API?
- A: Atovaquone should be stored in tightly sealed container111s at controlled room temperature (20-25°C), protected from light and moisture, to ensure product stability and shelf life.
Lead Time & Fulfillment
We maintain optimized inventory levels and efficient production schedules to ensure reliable and timely delivery of Atovaquone. Standard lead times typically range from 4 to 6 weeks, depending on order volume and specific customization requirements. For urgent orders or large-scale projects, we encourage early communication to facilitate expedited production and logistics planning. Our global logistics network ensures secure and compliant international shipping.
Warranty Commitments
We stand behind the quality of our Atovaquone API with a comprehensive warranty. Our product is guaranteed to meet all specified pharmacopoeial standards (USP, EP, BP) and agreed-upon customer specifications upon delivery and throughout its warranted shelf life, provided it is stored and handled according to our recommendations. Any deviation from these specifications will be addressed promptly and professionally in accordance with our stringent quality assurance protocols.
Customer Support & After-Sales Service
Our commitment extends beyond delivery. We offer dedicated technical support from experienced chemists and regulatory experts to assist with formulation development, analytical troubleshooting, and regulatory inquiries related to Atovaquone. Our customer service team is available to ensure smooth order processing, provide real-time updates, and resolve any post-delivery concerns. We believe in building long-term partnerships through unparalleled support.
Conclusion
The production and supply of high-quality Atovaquone API are crucial for addressing critical global health challenges such as malaria and opportunistic infections. Our unwavering commitment to excellence in manufacturing, stringent quality control, comprehensive regulatory support, and customer-centric service ensures that our partners receive an API that not only meets but often exceeds industry benchmarks. By leveraging our technical expertise and robust supply chain, pharmaceutical companies can confidently develop and deliver life-saving medications to patients worldwide.
References:
- United States Pharmacopeia. USP 43–NF 38. U.S. Pharmacopeial Convention; 2020.
- European Pharmacopoeia. Ph. Eur. 10.0. Council of Europe; 2020.
- World Health Organization. Guidelines for the Treatment of Malaria. 3rd ed. WHO Press; 2015.
- Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents. Department of Health and Human Services; 2023.
- ICH Harmonised Tripartite Guideline. Q7: Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use; 2000.
Hebei Guangxing Chemical Co., Ltd. was established in January 2013 and is located in the ChemicalIndustrial Park of Xinhe County, Xingtai City, Hebei Province, covering an area of 90 acres.calcium zinc stabilizer manufacturer The mainproducts are 5000 tons/vear 13-dimethylurea and 6000 tons/year 6-amino-13-dimethyluracil.Hebei Guangxing Chemical Co., Ltd. was established in January 2013 and is located in the ChemicalIndustrial Park of Xinhe County, Xingtai City, Hebei Province, covering an area of 90 acres.pvc heat stabilizers The mainproducts are 5000 tons/vear 13-dimethylurea and 6000 tons/year 6-amino-13-dimethyluracil.uracils|super blog